Healthcare technologies are rapidly evolving, producing new data types and innovative data uses. Data and technology can bring significant enhancements to the healthcare system, deepen patients’ and consumers’ engagement and understanding about their health, and be used as part of initiatives meant to improve health outcomes. It is critical to analyze how sensitive health and wellness data affect individual privacy and understand what it means for doctors, researchers, and companies to responsibly use such data. The FPF health team continues to build on its prior work on Consumer Wearables and Wellness Apps and Devices and Privacy Best Practices for Consumer Genetic Testing Services by exploring and addressing issues at the forefront and intersection of health, data, and privacy. Of main focus are the privacy challenges related to the collection, use, and sharing of both medical data and data that falls outside of the scope of HIPAA and FDA regulations. FPF brings together stakeholders to analyze how new technologies and data practices in the health ecosystem can impact individual privacy and promote the more effective and ethical use of data.
Featured
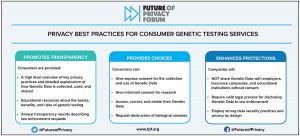
The DNA of Genetic Privacy Legislation: Montana, Tennessee, Texas, and Virginia Enter 2024 with New Genetic Privacy Laws Incorporating FPF’s Best Practices
In 2023, four states enacted new genetic privacy laws regulating direct-to-consumer genetic testing companies. This blog post provides details on what these new laws cover and how they compare to FPF’s widely-adopted Best Practices for Consumer Genetic Testing Services. Genetic privacy has been under increasing scrutiny at the state and federal levels, and regulators are […]
Regu(AI)ting Health: Lessons for Navigating the Complex Code of AI and Healthcare Regulations
Authors: Stephanie Wong, Amber Ezzell, & Felicity Slater As an increasing number of organizations utilize artificial intelligence (“AI”) in their patient-facing services, health organizations are seizing the opportunity to take advantage of the new wave of AI-powered tools. Policymakers, from United States (“U.S.”) government agencies to the White House, have taken heed of this trend, […]
FPF Files Comments for the FTC Health Breach Notification Rule Addressing Specific Definitions and Clarity of Scope
On August 8th, the Future of Privacy Forum (FPF) filed comments with the U.S. Federal Trade Commission (the Commission) regarding the Notice of Proposed Rulemaking (NPRM) to clarify the scope and application of the Health Breach Notification Rule (HBNR). The HBNR was promulgated in 2009 as part of the American Recovery and Reinvestment Act as […]
FPF Files Comments with the U.S. Department of Health and Human Services (HHS) Office for Civil Rights
On June 15, the Future of Privacy Forum (FPF) filed comments with the U.S. Department of Health and Human Services (HHS) Office for Civil Rights (OCR) regarding the Notice of Proposed Rulemaking (NPRM) on extending additional protections to reproductive health care data under the Health Insurance Portability and Accountability Act (HIPAA). One year ago last […]
(Health) Data is What (Health) Data Does in Nevada
Note: This title is inspired by Professor Daniel J. Solove’s recent essay, ‘Data Is What Data Does: Regulating Based on Harm and Risk Instead of Sensitive Data.’ On June 16, 2023, Nevada Senate Bill 370 (SB 370) was signed into law by Governor Lombardo, making Nevada the second state, after Washington, to pass broad-based consumer […]
Connecticut Shows You Can Have It All
On June 3rd, Connecticut Senate Bill 3 (SB 3), an “Act Concerning Online Privacy, Data and Safety Protections,” cleared the state legislature following unanimous votes in the House and Senate. If enacted by Governor Lamont, SB 3 will amend the Connecticut Data Privacy Act (CTDPA) to create new rights and protections for consumer health data […]
A New Paradigm for Consumer Health Data Privacy in Washington State
The Washington ‘My Health, My Data’ Act (MHMD or the Act) establishes a fundamentally new legal framework within U.S. law to regulate the collection, use, and transfer of consumer health data. Signed into law by Governor Inslee on April 27, MHMD was introduced by request of the Washington Attorney General in response to the Supreme […]
Five Big Questions (and Zero Predictions) for the U.S. State Privacy Landscape in 2023
Entering 2023, the United States remains one of the only global economic powers that lacks a comprehensive, national framework governing the collection and use of consumer data throughout the economy. Congress made unprecedented progress toward enacting baseline privacy legislation in 2022. However, the apparent impasse in the efforts to move H.R. 8152, the American Data […]
ITPI: New OECD-Israel Workshop January 2021 Report
The Israel Tech Policy Institute (ITPI) is excited to share a new report on key issues emerging from the OECD Workshop: ‘Supporting Health Innovation with Fair Information Practice Principles‘. The OECD Workshop was held virtually from 19 – 20 January 2021, in collaboration with the Israeli Ministry of Health and the Israel Tech Policy Institute. The report was drafted by Limor Shmerling […]
The Significance of Inclusion in Clinical Trials and Medical Research Databases
Our colleagues at the Israel Tech Policy Institute (ITPI) published a thoughtful blog on the significance of diversity and inclusion in clinical trials and health and medical research databases. They discuss the imperative of being represented in data, for one’s existence to be recognized and considered. When such data is the building block for a […]